Dr. Tautz develops novel drugs targeting protein tyrosine phosphatases that are implicated in cancer, thrombosis, and Alzheimer’s disease.
Dr. Tautz earned his PhD in Organic Chemistry and Biochemistry from the University of Karlsruhe (Germany) with Dr. Janos Retey in 2002. He continued his research at the Burnham Institute with Dr. Tomas Mustelin, first as a postdoc and later as a staff scientist. In 2009 Dr. Tautz joined the faculty of the Sanford Burnham Prebys Medical Discovery Institute.
Education
2002: PhD, University of Karlsruhe, Germany, Chemistry, Biochemistry
1997: MS, University of Karlsruhe, Germany, Chemistry
Funding Awards and Collaborative Grants
American Heart Association Innovative Research Grant
Honors and Recognition
2014: Semifinalist, Stadtman Investigator Search, National Institutes of Health
2008/2006 : Society for Biomolecular Sciences Travel Awards
2006: American Chemical Society Travel Award
2006: William and Lillian Fishman Award for Exceptional Postdoctoral Research
2002: PhD in Chemistry magna cum laude, University of Karlsruhe, Germany
Related Disease
Alzheimer’s Disease, Cancer, Cardiovascular Diseases, Inflammatory/Autoimmune Disease, Leukemia/Lymphoma, Neurodegenerative and Neuromuscular Diseases
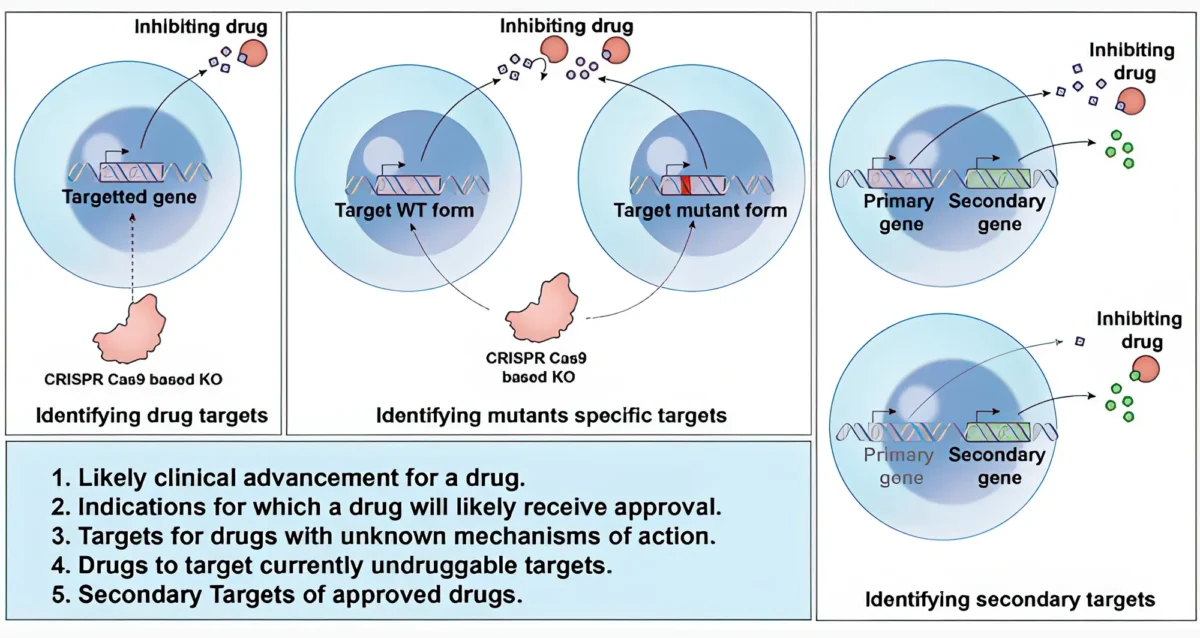
Many human diseases stem from abnormalities in the activities of protein kinases and protein phosphatases. While efficacious therapeutics targeting protein kinases have been successfully used in the clinic (e.g., Gleevec), effective strategies to target specific protein phosphatases are still elusive. Dr. Tautz’ laboratory works on novel, more effective approaches to target these important enzymes. He focuses on protein tyrosine phosphatases implicated in cancer, thrombosis, autoimmunity, and Alzheimer’s disease.
Lutz Tautz’s Research Report
1. Discovery of a Novel Drug Target in Arterial Thrombosis
Arterial thrombosis is the primary cause of most cases of myocardial infarction and stroke, the leading causes of death in the developed world. Platelets, highly specialized cells of the circulatory system, are key contributors to thrombotic events. Antiplatelet drugs, which prevent platelets from aggregating, have been very effective in reducing the mortality and morbidity of these conditions. However, approved antiplatelet therapies have adverse side effects, most notably the increased risk of bleeding. In collaboration with researchers at the University of Liege in Belgium (Drs. Souad Rahmouni and Cecile Oury), we recently identified DUSP3 (also known as VHR) as a major regulator in platelet signaling and thrombosis. Intriguingly, bleeding was not affected by DUSP3 deficiency in mice, suggesting that DUSP3 plays a key role in arterial thrombosis, but is dispensable for primary hemostasis. We develop a specific small-molecule inhibitor of DUSP3 that effectively inhibited platelet aggregation in human platelets, thereby phenocopying the effect of DUSP3 deficiency in murine platelets. We are now poised to optimize this compound for in vivo studies in order to provide proof-of-concept for a novel and potentially safer antiplatelet strategy based on DUSP3.
Publications:
- Dual-specificity phosphatase 3 deficiency or inhibition limits platelet activation and arterial thrombosis Musumeci L et al. Circulation. 2015 Feb 17;131(7):656-68.
- Perspective: Tyrosine phosphatases as novel targets for antiplatelet therapy. Tautz L et al. Bioorg Med Chem. 2015 Jun 15;23(12):2786-97.
2. Discovery of a Novel Mechanism in T Cell Activation
PTPs are crucial for maintaining the homeostasis of the immune system, including the regulation of antigen receptor-mediated lymphocyte activation and cytokine-induced differentiation. The lymphoid tyrosine phosphatase (LYP, PTPN22) is a critical negative regulator of T cell antigen receptor signaling. A single-nucleotide polymorphism (SNP) in PTPN22 was shown to correlate with the incidence of various autoimmune diseases, including type 1 diabetes and rheumatoid arthritis. First, we helped to demonstrate that the disease-associated allele is a gain-of-function mutant, i.e. a better inhibitor of T cell receptor activation. Then, we developed a specific chemical probe of LYP which we utilized to identify the associated mechanism that leads to increased LYP activity. In contrast to what was known from work in mice, we showed that in human cells LYP needs to dissociate from CSK in order to inhibit T cell activation. We also showed that our LYP inhibitor acts by stabilizing a unique inactive conformation of LYP.
Publications:
- Autoimmune-associated lymphoid tyrosine phosphatase is a gain-of-function variant. Vang T et al. Nat Genet. 2005 Dec;37(12):1317-9.
- In Silico Screening for PTPN22 Inhibitors: Active Hits From an Inactive Phosphatase Conformation. Wu S et al. ChemMedChem. 2009 Mar;4(3):440-4.
- Inhibition of Lymphoid Tyrosine Phosphatase by Benzofuran Salicylic Acids. Vang T et al. J Med Chem. 2011 Jan 27;54(2):562-71.
- LYP inhibits T cell activation when dissociated from CSK. Vang T et al. Nat Chem Biol. 2012 Mar 18;8(5):437-46.
3. Chemical Probe Development for Cancer Research
We have been working on chemical probes for cancer targets for several years (e.g., VHR, HePTP). Currently, we focus on the role of SHP2 in leukemia and breast cancer. PTPN11, the gene encoding SHP2, has been widely recognized as an oncogene. Germline mutations in PTPN11 were first observed in ~50% of cases of Noonan syndrome, an autosomal dominant developmental disorder with increased risk of malignancy. Numerous somatic gain-of-function mutations in PTPN11 have been identified in various leukemias. Hyperactivated SHP2 was also found in several types of solid tumors, including breast cancer. Previously reported SHP2 inhibitors lack efficacy in cancer cells and/or selectivity over related homologs. Novel SHP2 antagonists are needed for proof-of-principle studies that support a therapeutic approach based on SHP2. We have identified novel SHP2 lead compounds with significantly improved efficacy and selectivity. Currently, we optimize these compounds to make them suitable for in vivo studies.
Publications:
- Multidentate Small-Molecule Inhibitors of Vaccinia H1-Related (VHR) Phosphatase Decrease Proliferation of Cervix Cancer Cells. Wu S et al. J Med Chem. 2009 Nov 12;52(21):6716-23.
- Inhibition of the Hematopoietic Protein Tyrosine Phosphatase by Phenoxyacetic Acids. Bobkova EV et al. ACS Med Chem Lett. 2011 Feb;2(2):113-118.
- A Specific Inhibitor of Hematopoietic Protein Tyrosine Phosphatase Augments ERK1/2 and p38 Activation in vivo. Sergienko E et al. ACS Chem Biol. 2012 Feb 17; 7(2): 367–377.
- Protein tyrosine phosphatases: structure, function, and implication in human disease. Tautz L et al. Methods Mol Biol. 2013;1053:179-221.
4. Targeting STEP in Alzheimer’s Disease
Alzheimer’s disease (AD) is characterized by a progressive loss of cognitive function. The FDA has approved four drugs to treat the cognitive deficits in AD (donepezil, galantamine, rivastigmine, and memantine). However, none of these drugs halt disease progression. Our collaborator Dr. Paul Lombroso (Yale) identified the STriatal-Enriched Phosphatase (STEP) as a novel therapeutic target involved in the initial synaptic dysfunction that occurs prior to loss of neurons. His work suggests that inhibition of STEP could provide a disease-modifying strategy and early treatment option for AD. We recently received funding from the Alzheimer’s Association to develop a high-throughput assay to screen large chemical libraries for compounds that inhibit STEP function in neuronal cells. Once such compounds are identified, we will test their potential to reverse the biochemical and cognitive defects in AD animal models.
Publications:
- Inhibitor of the Tyrosine Phosphatase STEP Reverses Cognitive Deficits in a Mouse Model of Alzheimer’s Disease. Xu J et al. PLoS Biol. 2014 Aug 5;12(8):e1001923.