We caught up with cell biologist Anne Bang, who recently teamed up with her husband to study how SARS-CoV-2 affects the brain
Brain fog. Memory loss. Dizziness and confusion. Although COVID-19 is primarily thought of as a lung disease, survivors continue to report lingering and highly concerning neurological effects—severe enough to impact their ability to work and live normal lives. Doctors are also seeing a worrisome increase in strokes in younger patients, among other observations.
To learn what scientists know so far about COVID-19 and its effect on the brain, we caught up with Anne Bang, PhD, director of Cell Biology at Sanford Burnham Prebys’ Conrad Prebys Center for Chemical Genomics. Bang recently teamed up with scientists at Penn Medicine and a virologist at Scripps Research—who also happens to be her husband—to investigate whether SARS-CoV-2 infects brain cells. Their findings were published in Cell Stem Cell.
What do scientists know about the brain and COVID-19 so far?
Unfortunately, information is still very limited. There are reports of viral replication in the brain and spinal cord fluid of people with COVID-19 who have neurological symptoms. But as you can imagine, taking brain biopsies from someone who has COVID-19 is not realistic. So we really don’t know a lot yet. For this reason, scientists are turning to systems that can model the human brain, such as brain cells created from induced pluripotent stem cells (iPSCs) and brain organoids, to study SARS-CoV-2’s impact on the brain.
What did you find in your study?
We created several types of brain cells using iPSCs and brain organoids, which we then infected with SARS-CoV-2. We found that SARS-CoV-2 primarily infects a brain cell type called choroid plexus cells—largely bypassing neurons and astrocytes. The choroid plexus is a specialized part of the blood-brain barrier, which controls what can enter your brain and produces cerebral spinal fluid. More research emerges every day, but so far, the consensus in the field seems to align with our findings.
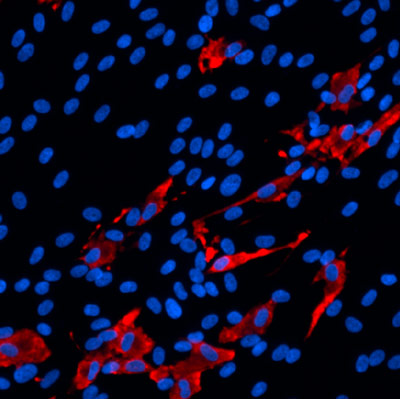
The scientists found that SARS-CoV-2 (red) primarily infects brain cells called choroid plexus cells (blue), which are part of the brain’s protective blood-brain barrier.
How might this finding translate to what we’re seeing in patients?
We know that choroid plexus cells produce high levels of ACE2, which is the receptor that SARS-CoV-2 uses to enter and infect cells. Because the choroid plexus is the “gatekeeper” to the brain, it’s possible that the virus enters the brain by infecting these cells. However, much more research is needed before we can give a definitive answer to this question.
We have more questions than answers right now about COVID-19. What is one question you wish we had the answer to?
How does the virus get from the nose and mouth and spread to other parts of the body? This is a big question for me and the scientific field. Once we know how the virus travels throughout the body, we can potentially stop its spread and control the dangerous symptoms.
What was it like working with your husband? Was this your first time working together?
It was really fun. I found out that he is great to work with. We’ve been together for 30 years, and incredibly, this was the first time we worked together.